ㆍPrivacy: We respect your privacy. Here you can find an example of a non-disclosure agreement. By submitting this form, you agree to our terms & conditions and privacy policy.
Views: 9 Author: Allen Xiao Publish Time: 2026-01-06 Origin: Site








In the medical device industry, there is no such thing as a "small mistake." A microscopic burr left on a bone screw can cause a devastating infection. A slight deviation in a dental implant's tolerance can prevent successful osseointegration. Manufacturing for the human body requires a mindset that goes far beyond standard engineering; it requires a reverence for life.
This is the domain of medical titanium precision machining. It is a field where rigorous validation, absolute traceability, and flawless surface finishes are not just requirements—they are moral obligations. At Jucheng Precision, we combine our advanced titanium cnc machining capabilities with a strict ISO 13485 quality management system to deliver components that surgeons trust with their patients' lives.
From material sourcing to sterile-ready packaging, this guide details how we maintain the highest standards of integrity for medical manufacturing.
content:

Titanium is not just a metal; in the medical world, it is a biomaterial. Its primary advantage is "Biocompatibility." The human immune system does not reject titanium. Instead, a phenomenon called Osseointegration occurs, where natural bone tissue actually grows into the microscopic pits of the titanium surface, locking the implant in place.
For medical titanium precision machining, we primarily use two specific grades:
Grade 2 CP: Used for non-load-bearing applications like surgical instrument handles and trays.
Grade 23 (Ti-6Al-4V ELI): "ELI" stands for Extra Low Interstitial. It is a purer version of Grade 5 with lower oxygen and iron content. This improves ductility and fracture toughness, making it the global standard for bone plates, screws, and hip replacements.

Many machine shops claim they can make medical parts, but without ISO 13485 certification, they represent a liability risk. Jucheng Precision is ISO 13485:2016 certified. This is not just a wall plaque; it is an operating system.
It means we practice proactive Risk Management (ISO 14971). Before we cut a single chip, we perform a PFMEA (Process Failure Mode and Effects Analysis) to identify what could go wrong. Could the coolant contaminate the surface? Could the heat treatment distort the thread? We identify these risks and implement controls to prevent them. When you audit Jucheng, you will find documented validation for every process, ensuring that the quality you get in the prototype phase is identical to the quality in mass production.

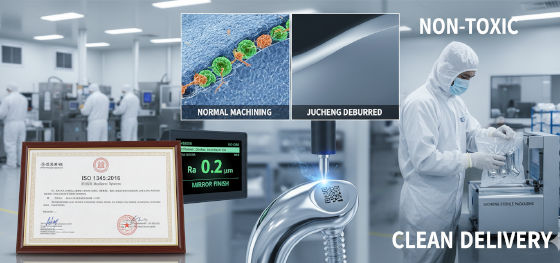
In automotive machining, a small burr might prevent a part from fitting. In medical machining, a small burr can detach inside a patient's body, leading to inflammation, infection, or blood clots. We operate under a strict "Zero-Burr Mandate."
Titanium is tough and tends to hold onto its burrs tenaciously. Standard tumbling is often not enough. Jucheng employs a multi-stage deburring strategy:
1. On-Machine Deburring: Using specialized micro-tools to chamfer edges inside the CNC cycle.
2. Thermal Deburring: For internal cross-holes.
3. Microscopic Inspection: Every critical medical component undergoes visual inspection under 10x to 40x magnification. If a burr is visible under the scope, the part is rejected.

Bacteria love rough surfaces. To prevent bacterial adhesion (biofilm formation), medical implants often require exceptionally smooth surface finishes, typically Ra 0.4 µm or better.
Jucheng Precision utilizes high-speed machining techniques and electropolishing to achieve mirror-like surfaces on titanium. Furthermore, "Cleanliness" refers to chemical purity. We use designated coolants free of sulfur and chlorine for medical runs to prevent surface contamination. After machining, parts undergo ultrasonic cleaning in a cleanroom environment to remove all traces of oil and particulate matter before packaging.

Imagine a scenario where a specific batch of titanium raw material was found to have a metallurgical defect. You need to know exactly which screws were made from that batch and where they are now. This is Traceability.
Jucheng maintains a "Golden Thread" of data. We track the Heat Number from the raw material mill certificate, link it to the Jucheng Lot Number, and verify it with in-house XRF testing. Finally, we use fiber lasers to engrave a UDI (Unique Device Identification) code—often a microscopic Data Matrix—directly onto the part. This allows hospitals to scan the device before surgery and pull up its entire manufacturing history.
Outsourcing medical parts is a leap of faith. You are trusting a supplier with your reputation and your patients' well-being.
Jucheng Precision honors this trust through transparency. We welcome audits. We provide full documentation packages (IQ/OQ/PQ assistance) upon request. Whether you are launching a new spinal fusion system or scaling up production of dental abutments, our ISO 13485 facility is ready to serve as your dedicated manufacturing partner. Contact us today to discuss your medical project with our engineering team.

